
We'll now focus on the different shapes caused by varying numbers of pairs of electrons, starting with molecules with just two pairs and working up to those with six. 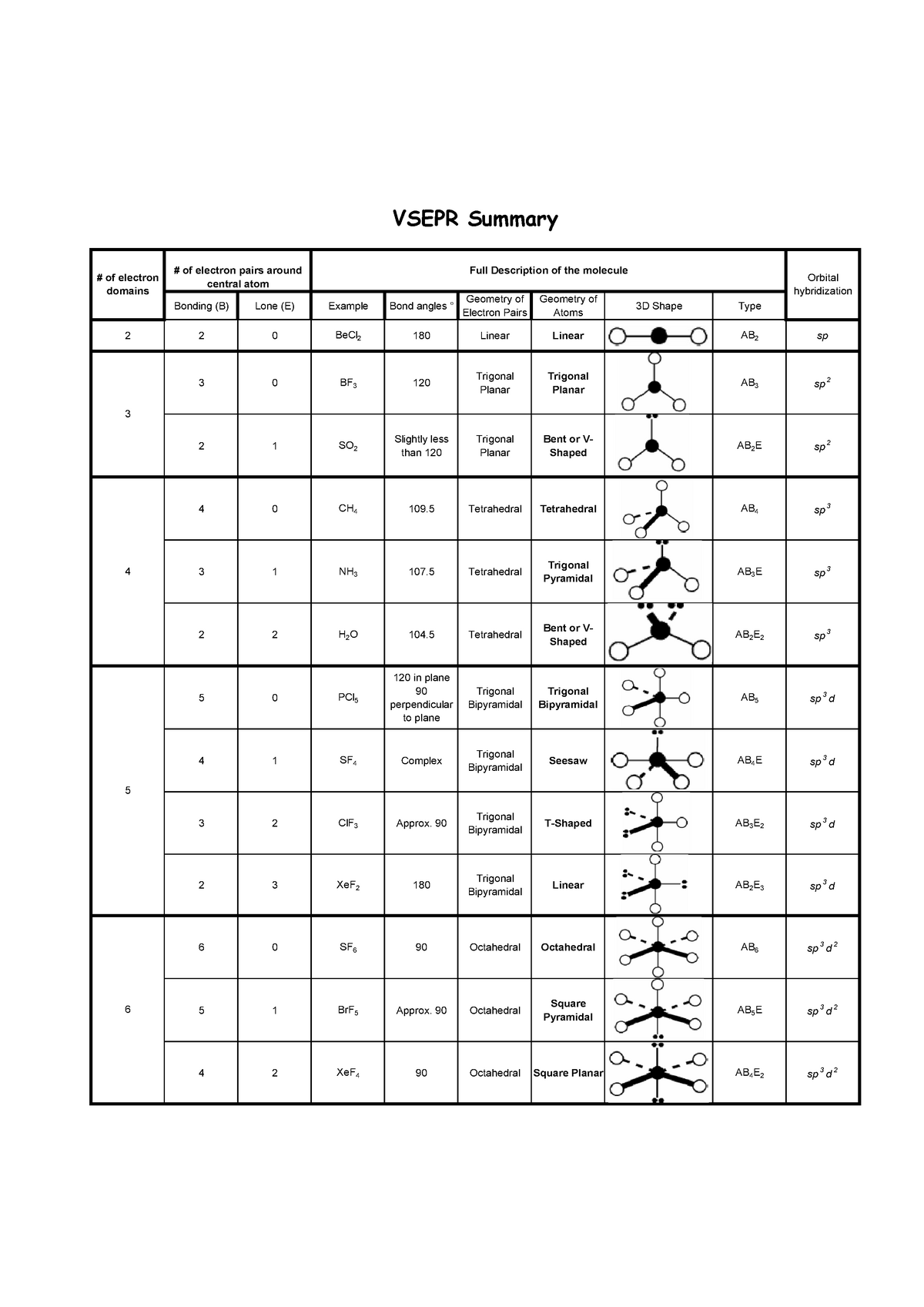
We've learned that VSEPR uses the number and arrangement of valence electron s to predict the geometry of a molecule. Now that we've looked at the fundamentals of VSEPR theory, let's move on to the shapes of the molecules themselves. Swap another bonded pair out, and the angle decreases down to 104.5 °, making the molecule v-shaped. But if you swap one of the bonded pairs for a lone pair, the angle between the three remaining bonds decreases down to 107.0 °, and the shape changes slightly, becoming trigonal pyramidal. If all four pairs of electrons are bonded pairs, the angle between the bonds is approximately 109.5 °. Source: Expiiįor example, a molecule with four pairs of electrons around a central atom is always based on a tetrahedral shape. This decreases the angle between the bonded pairs and changes the molecule's shape.Įxamples of molecules with lone and bonded pairs of electrons. If any lone pairs are present, they squeeze the bonded pairs closer together. This is because lone pairs of electrons repel other electron pairs much more strongly than bonded pairs. This alters the shape of the molecule ever so slightly. But when it comes to geometry and electron pair repulsion, not all electron pairs are equal. 
We now know that molecules with the same number of electron pairs all have the same basic shape. Because of this, the presence of lone electron pairs will squash two bonded electron pairs closer to each other, changing the geometry of the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
